| Weight | 1 lbs |
|---|---|
| Dimensions | 9 × 5 × 2 in |
| host | rabbit |
| isotype | IgG |
| clonality | polyclonal |
| concentration | 1 mg/mL |
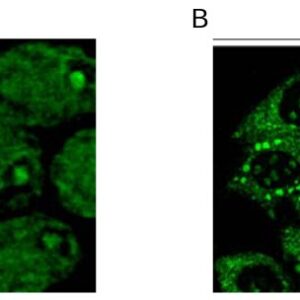
| applications | ICC/IF, WB |
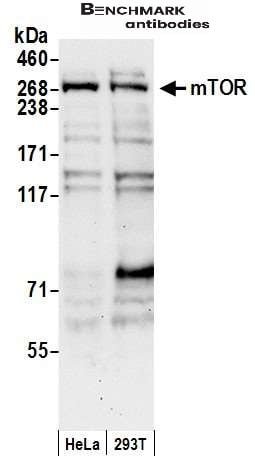
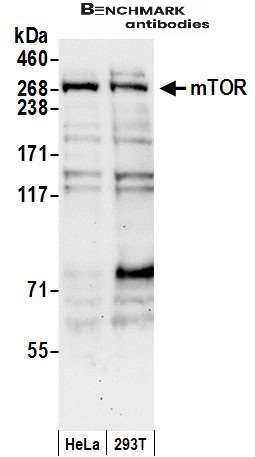
| reactivity | mTOR |
| available sizes | 100 µg |
rabbit anti-mTOR polyclonal antibody 4269
$518.00
Antibody summary
- Rabbit polyclonal to mTOR
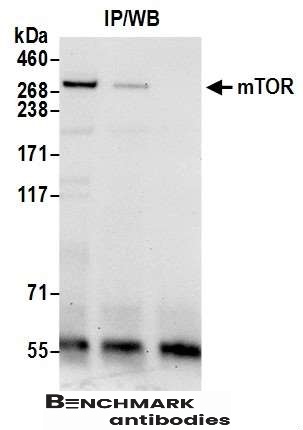
- Suitable for: WB,IP
- Isotype: Whole IgG
- 100 µg
rabbit anti-mTOR polyclonal antibody 4269
| antibody |
|---|
| Tested applications WB,IP |
| Recommended dilutions Western Blot: 0.1-1 ug/ml.Note: Overnight incubation at 4°C is recommended for primary antibody. MTOR requires detergent and extended Western Blotting transfer times (e.g. in 5% acrylamide gels to PVDF, use 50V for 3 hours in CAPS buffer with 10% methanol and 0.1% SDS). Immunoprecipitati |
| Immunogen Synthetic peptide representing a portion of the protein encoded in part by exons 5 and 6 (LocusLink ID 1981). |
| Size and concentration 100µg and lot specific |
| Form liquid |
| Storage Instructions Store at 2 - 8°C. Antibody is stable at 2 - 8°C for 1 year. |
| Storage buffer Tris-citrate/phosphate buffer, pH 7 to 8 contai |
| Purity immunogen affinity purification |
| Clonality polyclonal |
| Isotype IgG |
| Compatible secondaries goat anti-rabbit IgG, H&L chain specific, peroxidase conjugated, conjugated polyclonal antibody 9512 goat anti-rabbit IgG, H&L chain specific, biotin conjugated polyclonal antibody 2079 goat anti-rabbit IgG, H&L chain specific, FITC conjugated polyclonal antibody 7863 goat anti-rabbit IgG, H&L chain specific, Cross Absorbed polyclonal antibody 2371 goat anti-rabbit IgG, H&L chain specific, biotin conjugated polyclonal antibody, crossabsorbed 1715 goat anti-rabbit IgG, H&L chain specific, FITC conjugated polyclonal antibody, crossabsorbed 1720 |
| Isotype control Rabbit polyclonal - Isotype Control |
| target relevance |
|---|
| Protein names Serine/threonine-protein kinase mTOR (EC 2.7.11.1) (FK506-binding protein 12-rapamycin complex-associated protein 1) (FKBP12-rapamycin complex-associated protein) (Mammalian target of rapamycin) (mTOR) (Mechanistic target of rapamycin) (Rapamycin and FKBP12 target 1) (Rapamycin target protein 1) (Tyrosine-protein kinase mTOR) (EC 2.7.10.2) |
| Gene names MTOR,MTOR FRAP FRAP1 FRAP2 RAFT1 RAPT1 |
| Protein family PI3/PI4-kinase family |
| Mass 288892Da |
| Function FUNCTION: Serine/threonine protein kinase which is a central regulator of cellular metabolism, growth and survival in response to hormones, growth factors, nutrients, energy and stress signals (PubMed:12087098, PubMed:12150925, PubMed:12150926, PubMed:12231510, PubMed:12718876, PubMed:14651849, PubMed:15268862, PubMed:15467718, PubMed:15545625, PubMed:15718470, PubMed:18497260, PubMed:18762023, PubMed:18925875, PubMed:20516213, PubMed:20537536, PubMed:21659604, PubMed:23429703, PubMed:23429704, PubMed:25799227, PubMed:26018084, PubMed:29150432, PubMed:29236692, PubMed:31112131, PubMed:31601708, PubMed:32561715, PubMed:34519269, PubMed:37751742). MTOR directly or indirectly regulates the phosphorylation of at least 800 proteins (PubMed:15268862, PubMed:15467718, PubMed:17517883, PubMed:18372248, PubMed:18497260, PubMed:18925875, PubMed:20516213, PubMed:21576368, PubMed:21659604, PubMed:23429704, PubMed:29236692, PubMed:37751742). Functions as part of 2 structurally and functionally distinct signaling complexes mTORC1 and mTORC2 (mTOR complex 1 and 2) (PubMed:15268862, PubMed:15467718, PubMed:18497260, PubMed:18925875, PubMed:20516213, PubMed:21576368, PubMed:21659604, PubMed:23429704, PubMed:29424687, PubMed:29567957, PubMed:35926713). In response to nutrients, growth factors or amino acids, mTORC1 is recruited to the lysosome membrane and promotes protein, lipid and nucleotide synthesis by phosphorylating key regulators of mRNA translation and ribosome synthesis (PubMed:12087098, PubMed:12150925, PubMed:12150926, PubMed:12231510, PubMed:12718876, PubMed:14651849, PubMed:15268862, PubMed:15467718, PubMed:15545625, PubMed:15718470, PubMed:18497260, PubMed:18762023, PubMed:18925875, PubMed:20516213, PubMed:20537536, PubMed:21659604, PubMed:23429703, PubMed:23429704, PubMed:25799227, PubMed:26018084, PubMed:29150432, PubMed:29236692, PubMed:31112131, PubMed:34519269). This includes phosphorylation of EIF4EBP1 and release of its inhibition toward the elongation initiation factor 4E (eiF4E) (PubMed:24403073, PubMed:29236692). Moreover, phosphorylates and activates RPS6KB1 and RPS6KB2 that promote protein synthesis by modulating the activity of their downstream targets including ribosomal protein S6, eukaryotic translation initiation factor EIF4B, and the inhibitor of translation initiation PDCD4 (PubMed:12087098, PubMed:12150925, PubMed:18925875, PubMed:29150432, PubMed:29236692). Stimulates the pyrimidine biosynthesis pathway, both by acute regulation through RPS6KB1-mediated phosphorylation of the biosynthetic enzyme CAD, and delayed regulation, through transcriptional enhancement of the pentose phosphate pathway which produces 5-phosphoribosyl-1-pyrophosphate (PRPP), an allosteric activator of CAD at a later step in synthesis, this function is dependent on the mTORC1 complex (PubMed:23429703, PubMed:23429704). Regulates ribosome synthesis by activating RNA polymerase III-dependent transcription through phosphorylation and inhibition of MAF1 an RNA polymerase III-repressor (PubMed:20516213). Activates dormant ribosomes by mediating phosphorylation of SERBP1, leading to SERBP1 inactivation and reactivation of translation (PubMed:36691768). In parallel to protein synthesis, also regulates lipid synthesis through SREBF1/SREBP1 and LPIN1 (PubMed:23426360). To maintain energy homeostasis mTORC1 may also regulate mitochondrial biogenesis through regulation of PPARGC1A (By similarity). In the same time, mTORC1 inhibits catabolic pathways: negatively regulates autophagy through phosphorylation of ULK1 (PubMed:32561715). Under nutrient sufficiency, phosphorylates ULK1 at 'Ser-758', disrupting the interaction with AMPK and preventing activation of ULK1 (PubMed:32561715). Also prevents autophagy through phosphorylation of the autophagy inhibitor DAP (PubMed:20537536). Also prevents autophagy by phosphorylating RUBCNL/Pacer under nutrient-rich conditions (PubMed:30704899). Prevents autophagy by mediating phosphorylation of AMBRA1, thereby inhibiting AMBRA1 ability to mediate ubiquitination of ULK1 and interaction between AMBRA1 and PPP2CA (PubMed:23524951, PubMed:25438055). mTORC1 exerts a feedback control on upstream growth factor signaling that includes phosphorylation and activation of GRB10 a INSR-dependent signaling suppressor (PubMed:21659604). Among other potential targets mTORC1 may phosphorylate CLIP1 and regulate microtubules (PubMed:12231510). The mTORC1 complex is inhibited in response to starvation and amino acid depletion (PubMed:12150925, PubMed:12150926, PubMed:24403073, PubMed:31695197). The non-canonical mTORC1 complex, which acts independently of RHEB, specifically mediates phosphorylation of MiT/TFE factors MITF, TFEB and TFE3 in the presence of nutrients, promoting their cytosolic retention and inactivation (PubMed:22343943, PubMed:22576015, PubMed:22692423, PubMed:24448649, PubMed:32612235, PubMed:36608670, PubMed:36697823). Upon starvation or lysosomal stress, inhibition of mTORC1 induces dephosphorylation and nuclear translocation of TFEB and TFE3, promoting their transcription factor activity (PubMed:22343943, PubMed:22576015, PubMed:22692423, PubMed:24448649, PubMed:32612235, PubMed:36608670). The mTORC1 complex regulates pyroptosis in macrophages by promoting GSDMD oligomerization (PubMed:34289345). MTOR phosphorylates RPTOR which in turn inhibits mTORC1 (By similarity). As part of the mTORC2 complex, MTOR transduces signals from growth factors to pathways involved in proliferation, cytoskeletal organization, lipogenesis and anabolic output (PubMed:15268862, PubMed:15467718, PubMed:24670654, PubMed:29424687, PubMed:29567957, PubMed:35926713). In response to growth factors, mTORC2 phosphorylates and activates AGC protein kinase family members, including AKT (AKT1, AKT2 and AKT3), PKC (PRKCA, PRKCB and PRKCE) and SGK1 (PubMed:15268862, PubMed:15467718, PubMed:21376236, PubMed:24670654, PubMed:29424687, PubMed:29567957, PubMed:35926713). In contrast to mTORC1, mTORC2 is nutrient-insensitive (PubMed:15467718). mTORC2 plays a critical role in AKT1 activation by mediating phosphorylation of different sites depending on the context, such as 'Thr-450', 'Ser-473', 'Ser-477' or 'Thr-479', facilitating the phosphorylation of the activation loop of AKT1 on 'Thr-308' by PDPK1/PDK1 which is a prerequisite for full activation (PubMed:15718470, PubMed:21376236, PubMed:24670654, PubMed:29424687, PubMed:29567957). mTORC2 also regulates the phosphorylation of SGK1 at 'Ser-422' (PubMed:18925875). mTORC2 may regulate the actin cytoskeleton, through phosphorylation of PRKCA, PXN and activation of the Rho-type guanine nucleotide exchange factors RHOA and RAC1A or RAC1B (PubMed:15268862). The mTORC2 complex also phosphorylates various proteins involved in insulin signaling, such as FBXW8 and IGF2BP1 (By similarity). May also regulate insulin signaling by acting as a tyrosine protein kinase that catalyzes phosphorylation of IGF1R and INSR; additional evidence are however required to confirm this result in vivo (PubMed:26584640). Regulates osteoclastogenesis by adjusting the expression of CEBPB isoforms (By similarity). Plays an important regulatory role in the circadian clock function; regulates period length and rhythm amplitude of the suprachiasmatic nucleus (SCN) and liver clocks (By similarity). {ECO:0000250|UniProtKB:Q9JLN9, ECO:0000269|PubMed:12087098, ECO:0000269|PubMed:12150925, ECO:0000269|PubMed:12150926, ECO:0000269|PubMed:12231510, ECO:0000269|PubMed:12718876, ECO:0000269|PubMed:14651849, ECO:0000269|PubMed:15268862, ECO:0000269|PubMed:15467718, ECO:0000269|PubMed:15545625, ECO:0000269|PubMed:15718470, ECO:0000269|PubMed:17517883, ECO:0000269|PubMed:18372248, ECO:0000269|PubMed:18497260, ECO:0000269|PubMed:18762023, ECO:0000269|PubMed:18925875, ECO:0000269|PubMed:20516213, ECO:0000269|PubMed:20537536, ECO:0000269|PubMed:21376236, ECO:0000269|PubMed:21576368, ECO:0000269|PubMed:21659604, ECO:0000269|PubMed:22343943, ECO:0000269|PubMed:22576015, ECO:0000269|PubMed:22692423, ECO:0000269|PubMed:23426360, ECO:0000269|PubMed:23429703, ECO:0000269|PubMed:23429704, ECO:0000269|PubMed:23524951, ECO:0000269|PubMed:24403073, ECO:0000269|PubMed:24448649, ECO:0000269|PubMed:24670654, ECO:0000269|PubMed:25438055, ECO:0000269|PubMed:25799227, ECO:0000269|PubMed:26018084, ECO:0000269|PubMed:26584640, ECO:0000269|PubMed:29150432, ECO:0000269|PubMed:29236692, ECO:0000269|PubMed:29424687, ECO:0000269|PubMed:29567957, ECO:0000269|PubMed:30704899, ECO:0000269|PubMed:31112131, ECO:0000269|PubMed:31601708, ECO:0000269|PubMed:31695197, ECO:0000269|PubMed:32561715, ECO:0000269|PubMed:32612235, ECO:0000269|PubMed:34289345, ECO:0000269|PubMed:34519269, ECO:0000269|PubMed:35926713, ECO:0000269|PubMed:36608670, ECO:0000269|PubMed:36691768, ECO:0000269|PubMed:36697823, ECO:0000269|PubMed:37751742}. |
| Catalytic activity CATALYTIC ACTIVITY: Reaction=L-seryl-[protein] + ATP = O-phospho-L-seryl-[protein] + ADP + H(+); Xref=Rhea:RHEA:17989, Rhea:RHEA-COMP:9863, Rhea:RHEA-COMP:11604, ChEBI:CHEBI:15378, ChEBI:CHEBI:29999, ChEBI:CHEBI:30616, ChEBI:CHEBI:83421, ChEBI:CHEBI:456216; EC=2.7.11.1; Evidence={ECO:0000269|PubMed:12087098, ECO:0000269|PubMed:12150925, ECO:0000269|PubMed:12231510, ECO:0000269|PubMed:15268862, ECO:0000269|PubMed:15467718, ECO:0000269|PubMed:15718470, ECO:0000269|PubMed:17517883, ECO:0000269|PubMed:18372248, ECO:0000269|PubMed:18925875, ECO:0000269|PubMed:20022946, ECO:0000269|PubMed:20516213, ECO:0000269|PubMed:20537536, ECO:0000269|PubMed:21659604, ECO:0000269|PubMed:22343943, ECO:0000269|PubMed:22576015, ECO:0000269|PubMed:22692423, ECO:0000269|PubMed:29150432}; CATALYTIC ACTIVITY: Reaction=L-threonyl-[protein] + ATP = O-phospho-L-threonyl-[protein] + ADP + H(+); Xref=Rhea:RHEA:46608, Rhea:RHEA-COMP:11060, Rhea:RHEA-COMP:11605, ChEBI:CHEBI:15378, ChEBI:CHEBI:30013, ChEBI:CHEBI:30616, ChEBI:CHEBI:61977, ChEBI:CHEBI:456216; EC=2.7.11.1; Evidence={ECO:0000269|PubMed:12087098, ECO:0000269|PubMed:12150925, ECO:0000269|PubMed:12231510, ECO:0000269|PubMed:15268862, ECO:0000269|PubMed:15467718, ECO:0000269|PubMed:15718470, ECO:0000269|PubMed:18925875, ECO:0000269|PubMed:20516213, ECO:0000269|PubMed:20537536, ECO:0000269|PubMed:21659604, ECO:0000269|PubMed:29150432, ECO:0000269|PubMed:29236692, ECO:0000269|PubMed:37751742}; CATALYTIC ACTIVITY: Reaction=L-tyrosyl-[protein] + ATP = O-phospho-L-tyrosyl-[protein] + ADP + H(+); Xref=Rhea:RHEA:10596, Rhea:RHEA-COMP:10136, Rhea:RHEA-COMP:20101, ChEBI:CHEBI:15378, ChEBI:CHEBI:30616, ChEBI:CHEBI:46858, ChEBI:CHEBI:61978, ChEBI:CHEBI:456216; EC=2.7.10.2; Evidence={ECO:0000305|PubMed:26584640}; PhysiologicalDirection=left-to-right; Xref=Rhea:RHEA:10597; Evidence={ECO:0000305|PubMed:26584640}; |
| Subellular location SUBCELLULAR LOCATION: Lysosome membrane {ECO:0000269|PubMed:18497260, ECO:0000269|PubMed:20381137, ECO:0000269|PubMed:29750193, ECO:0000269|PubMed:30956113, ECO:0000269|PubMed:31112131, ECO:0000269|PubMed:31601764, ECO:0000269|PubMed:37057673}; Peripheral membrane protein {ECO:0000269|PubMed:30956113, ECO:0000269|PubMed:31112131}; Cytoplasmic side {ECO:0000269|PubMed:30956113, ECO:0000269|PubMed:31112131}. Endoplasmic reticulum membrane {ECO:0000269|PubMed:14578359}; Peripheral membrane protein {ECO:0000269|PubMed:14578359}; Cytoplasmic side {ECO:0000269|PubMed:14578359}. Golgi apparatus membrane {ECO:0000269|PubMed:14578359}; Peripheral membrane protein {ECO:0000269|PubMed:14578359}; Cytoplasmic side {ECO:0000269|PubMed:14578359}. Cell membrane {ECO:0000269|PubMed:21867682}; Peripheral membrane protein {ECO:0000269|PubMed:14578359}. Mitochondrion outer membrane {ECO:0000269|PubMed:11930000, ECO:0000269|PubMed:14578359}; Peripheral membrane protein {ECO:0000269|PubMed:11930000, ECO:0000269|PubMed:14578359}; Cytoplasmic side {ECO:0000269|PubMed:11930000, ECO:0000269|PubMed:14578359}. Cytoplasm {ECO:0000269|PubMed:11930000, ECO:0000269|PubMed:18497260}. Nucleus {ECO:0000250|UniProtKB:Q9JLN9}. Nucleus, PML body {ECO:0000250|UniProtKB:Q9JLN9}. Microsome membrane {ECO:0000269|PubMed:9434772}. Cytoplasmic vesicle, phagosome {ECO:0000269|PubMed:27623384}. Note=Shuttles between cytoplasm and nucleus. Accumulates in the nucleus in response to hypoxia (By similarity). Targeting to lysosomes depends on amino acid availability and RRAGA and RRAGB (PubMed:18497260, PubMed:20381137). Lysosome targeting also depends on interaction with MEAK7. Translocates to the lysosome membrane in the presence of TM4SF5 (PubMed:30956113). The mTORC2 complex localizes to membranes: mTORC2 is active at the plasma membrane, endoplasmic reticulum membrane and lysosomes (PubMed:21867682). {ECO:0000250|UniProtKB:Q9JLN9, ECO:0000269|PubMed:18497260, ECO:0000269|PubMed:20381137, ECO:0000269|PubMed:21867682, ECO:0000269|PubMed:29750193, ECO:0000269|PubMed:30956113}. |
| Tissues TISSUE SPECIFICITY: Expressed in numerous tissues, with highest levels in testis. {ECO:0000269|PubMed:12408816, ECO:0000269|PubMed:7809080}. |
| Structure SUBUNIT: Part of the mechanistic target of rapamycin complex 1 (mTORC1) which contains MTOR, MLST8 and RPTOR (PubMed:12150925, PubMed:12150926, PubMed:12408816, PubMed:12718876, PubMed:18925875, PubMed:20542007, PubMed:23636326, PubMed:24403073, PubMed:26678875, PubMed:27909983, PubMed:29236692, PubMed:31601764, PubMed:34519268, PubMed:34519269, PubMed:36697823). The mTORC1 complex is a 1 Md obligate dimer of two stoichiometric heterotetramers with overall dimensions of 290 A x 210 A x 135 A (PubMed:20542007, PubMed:23636326). It has a rhomboid shape and a central cavity, the dimeric interfaces are formed by interlocking interactions between the two MTOR and the two RPTOR subunits (PubMed:20542007, PubMed:23636326, PubMed:27909983). The MLST8 subunit forms distal foot-like protuberances, and contacts only one MTOR within the complex, while the small AKT1S1/PRAS40 localizes to the midsection of the central core, in close proximity to RPTOR (PubMed:20542007, PubMed:23636326, PubMed:27909983, PubMed:29236692). mTORC1 associates with AKT1S1/PRAS40, which inhibits its activity by blocking MTOR substrate-recruitment site (PubMed:17386266, PubMed:29236692). Component of the mechanistic target of rapamycin complex 2 (mTORC2), consisting in two heterotretramers composed of MTOR, MLST8, RICTOR and MAPKAP1/SIN1 (PubMed:15268862, PubMed:15467718, PubMed:17599906, PubMed:18925875, PubMed:29424687, PubMed:29567957, PubMed:33158864, PubMed:35926713). Interacts with PLPP7 and PML (By similarity). Interacts with PRR5 and RICTOR; the interaction is direct within the mTORC2 complex and interaction with RICTOR is enhanced by deubiquitination of RICTOR by USP9X (PubMed:17599906, PubMed:33378666, PubMed:29424687, PubMed:34519268). mTORC1 and mTORC2 associate with DEPTOR, which regulates their activity (PubMed:19446321, PubMed:34519268, PubMed:34519269). Interacts with WAC; WAC positively regulates MTOR activity by promoting the assembly of the TTT complex composed of TELO2, TTI1 and TTI2 and the RUVBL complex composed of RUVBL1 and RUVBL2 into the TTT-RUVBL complex which leads to the dimerization of the mTORC1 complex and its subsequent activation (PubMed:26812014). Interacts with UBQLN1 (PubMed:11853878). Interacts with TTI1 and TELO2 (PubMed:20427287, PubMed:20801936, PubMed:20810650). Interacts with CLIP1; phosphorylates and regulates CLIP1 (PubMed:12231510). Interacts with NBN (PubMed:23762398). Interacts with HTR6 (PubMed:23027611). Interacts with BRAT1 (PubMed:25657994). Interacts with MEAK7 (via C-terminal domain); the interaction increases upon nutrient stimulation (PubMed:29750193). Interacts with TM4SF5; the interaction is positively regulated by arginine and is negatively regulated by leucine (PubMed:30956113). Interacts with GPR137B (PubMed:31036939). Interacts with NCKAP1L (PubMed:32647003). Interacts with TPCN1 and TPCN2; the interaction is required for TPCN1 and TPCN2 sensitivity to ATP (PubMed:23394946). Interacts with ATP6V1A and with CRYAB, forming a ternary complex (By similarity). Interacts with SLC38A7; this interaction mediates the recruitment of mTORC1 to the lysosome and its subsequent activation (PubMed:35561222). Interacts with TSPAN8 (PubMed:35904232). {ECO:0000250|UniProtKB:Q9JLN9, ECO:0000269|PubMed:11853878, ECO:0000269|PubMed:12150925, ECO:0000269|PubMed:12150926, ECO:0000269|PubMed:12231510, ECO:0000269|PubMed:12408816, ECO:0000269|PubMed:12718876, ECO:0000269|PubMed:15268862, ECO:0000269|PubMed:15467718, ECO:0000269|PubMed:17386266, ECO:0000269|PubMed:17599906, ECO:0000269|PubMed:18925875, ECO:0000269|PubMed:19446321, ECO:0000269|PubMed:20427287, ECO:0000269|PubMed:20542007, ECO:0000269|PubMed:20801936, ECO:0000269|PubMed:20810650, ECO:0000269|PubMed:23027611, ECO:0000269|PubMed:23394946, ECO:0000269|PubMed:23636326, ECO:0000269|PubMed:23762398, ECO:0000269|PubMed:24403073, ECO:0000269|PubMed:25657994, ECO:0000269|PubMed:26678875, ECO:0000269|PubMed:26812014, ECO:0000269|PubMed:27909983, ECO:0000269|PubMed:29236692, ECO:0000269|PubMed:29424687, ECO:0000269|PubMed:29567957, ECO:0000269|PubMed:29750193, ECO:0000269|PubMed:30956113, ECO:0000269|PubMed:31036939, ECO:0000269|PubMed:31601764, ECO:0000269|PubMed:32647003, ECO:0000269|PubMed:33158864, ECO:0000269|PubMed:33378666, ECO:0000269|PubMed:34519268, ECO:0000269|PubMed:34519269, ECO:0000269|PubMed:35561222, ECO:0000269|PubMed:35904232, ECO:0000269|PubMed:35926713, ECO:0000269|PubMed:36697823}. |
| Post-translational modification PTM: Autophosphorylates when part of mTORC1 or mTORC2 (PubMed:15467718, PubMed:20022946, PubMed:9434772). Phosphorylation at Ser-1261, Ser-2159 and Thr-2164 promotes autophosphorylation (PubMed:19487463). Phosphorylated at Ser-2448 by RPS6KB1 (PubMed:15899889, PubMed:15905173, PubMed:19145465). Phosphorylation in the kinase domain modulates the interactions of MTOR with RPTOR and AKT1S1/PRAS40 and leads to increased intrinsic mTORC1 kinase activity (PubMed:15905173, PubMed:19145465, PubMed:21576368). Phosphorylation at Ser-2159 by TBK1 in response to growth factors and pathogen recognition receptors promotes mTORC1 activity (PubMed:29150432). Phosphorylation at Ser-2159 by TBK1 in response to EGF growth factor promotes mTORC2 activity, leading to AKT1 phosphorylation and activation (By similarity). Phosphorylation at Thr-2173 in the ATP-binding region by AKT1 strongly reduces kinase activity (PubMed:24247430). {ECO:0000250|UniProtKB:Q9JLN9, ECO:0000269|PubMed:15467718, ECO:0000269|PubMed:15905173, ECO:0000269|PubMed:19145465, ECO:0000269|PubMed:19487463, ECO:0000269|PubMed:20022946, ECO:0000269|PubMed:21576368, ECO:0000269|PubMed:24247430, ECO:0000269|PubMed:29150432, ECO:0000269|PubMed:9434772}.; PTM: Ubiquitinated at Lys-2066 by the SCF(FBXO22) complex via 'Lys-27'-linked ubiquitination prevents mTORC1 substrate recruitment. {ECO:0000269|PubMed:37979583}. |
| Domain DOMAIN: The kinase domain (PI3K/PI4K) is intrinsically active but has a highly restricted catalytic center. {ECO:0000269|PubMed:23636326}.; DOMAIN: The FAT domain forms three discontinuous subdomains of alpha-helical TPR repeats plus a single subdomain of HEAT repeats. The four domains pack sequentially to form a C-shaped a-solenoid that clamps onto the kinase domain (PubMed:23636326). {ECO:0000269|PubMed:23636326}. |
| Involvement in disease DISEASE: Smith-Kingsmore syndrome (SKS) [MIM:616638]: An autosomal dominant syndrome characterized by intellectual disability, macrocephaly, seizures, umbilical hernia, and facial dysmorphic features. {ECO:0000269|PubMed:25851998, ECO:0000269|PubMed:26542245, ECO:0000269|PubMed:27830187}. Note=The disease is caused by variants affecting the gene represented in this entry.; DISEASE: Focal cortical dysplasia 2 (FCORD2) [MIM:607341]: A form of focal cortical dysplasia, a malformation of cortical development that results in medically refractory epilepsy in the pediatric population and in adults. FCORD2 is a severe form, with onset usually in childhood, characterized by disrupted cortical lamination and specific cytological abnormalities. It is classified in 2 subtypes: type IIA characterized by dysmorphic neurons and lack of balloon cells; type IIB with dysmorphic neurons and balloon cells. {ECO:0000269|PubMed:25799227, ECO:0000269|PubMed:25878179, ECO:0000269|PubMed:26018084, ECO:0000269|PubMed:27830187}. Note=The disease is caused by variants affecting the gene represented in this entry. |
| Target Relevance information above includes information from UniProt accession: P42345 |
| The UniProt Consortium |
Data
Publications
| pmid | title | authors | citation |
|---|---|---|---|
| We haven't added any publications to our database yet. | |||
Protocols
| relevant to this product |
|---|
| Western blot |
Documents
| # | SDS | Certificate | |
|---|---|---|---|
| Please enter your product and batch number here to retrieve product datasheet, SDS, and QC information. | |||
Only logged in customers who have purchased this product may leave a review.




Reviews
There are no reviews yet.