Showing the single result
Beta Galactosidase antibodies
Beta-galactosidase (?-gal) is a well-known enzyme commonly used in molecular biology, particularly as a reporter in gene expression studies. It hydrolyzes lactose into glucose and galactose and is naturally found in organisms such as Escherichia coli. In research, ?-gal is often fused to proteins of interest to track their expression and localization, due to its stability and measurable enzymatic activity.
In Western blotting (WB), ?-gal antibodies are used to detect ?-galactosidase-tagged proteins, providing an efficient means of tracking protein expression in experimental samples. These antibodies are highly specific, making them suitable for WB, immunofluorescence (IF), and immunohistochemistry (IHC) applications. The ?-gal antibodies available on your website can help ensure reliable detection and quantification of ?-gal-tagged proteins, enabling researchers to validate expression and monitor protein localization in various systems.
Beyond its use as a reporter, ?-gal also has roles in metabolism and cellular function, further expanding its significance in research.
| target | type | reactivity | applications | ||||
|---|---|---|---|---|---|---|---|
| 1779 | BGAL | chicken polyclonal | other | WB ICC IHC | |||

WB with antibody [1779] : Immunofluorescence cell staining- chicken anti-Beta Galactosidase polyclonal antibody 1779
Immunofluorescent analysis of 4% paraformaldehyde (PFA) - fixed, permeabilized with 0.1% triton X-100 CHO cells transfected with beta galactosidase constructs (CHO-BGAL) labeling beta galactosidase with antibody #1779 at 1/1000 dilution (1µg/mL), followed by Donkey Anti-chicken IgY, Alexa 594 conjugate at 1/1000 (1µg/mL) (red). Nucleus was counterstained with DAPI (blue). Also shown staining in parental cell line.
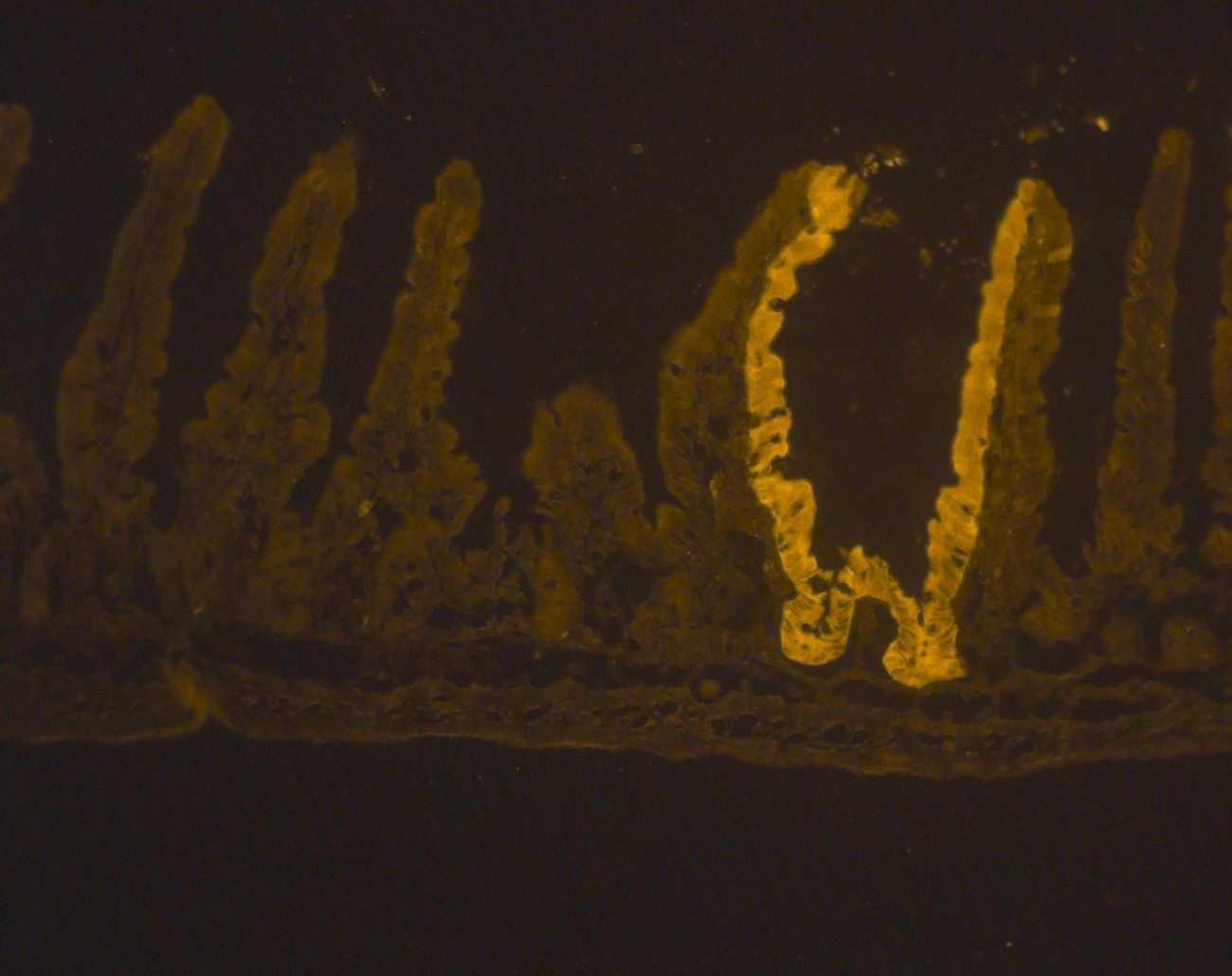
IHC with antibody [1779] : IHC cell staining- chicken anti-Beta Galactosidase polyclonal antibody 1779
